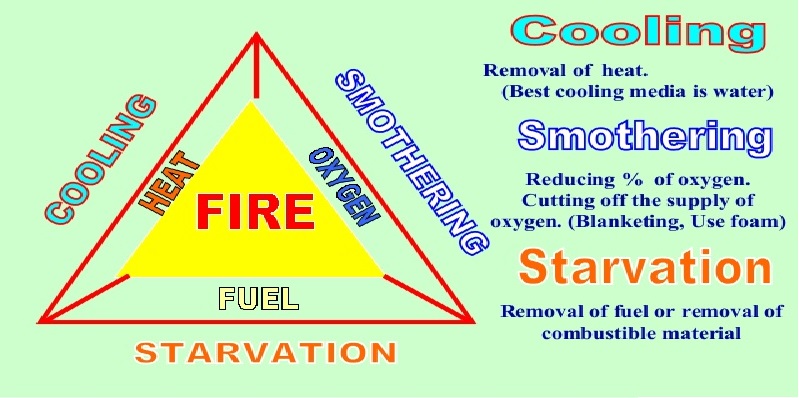
To understand how best to extinguish a fire, there must first be an appreciation of the three elements that make up the ‘fire triangle’: heat, fuel and oxygen. As fire is primarily a chemical reaction, removing one or more of these three factors will prevent the combustion from taking place.
Heat is require d to ignite a fire, and will continue to be generated as the fire burns. For intentional fires, this could
be as simple as the striking of a match. For accidental fires however, ignition can occur as a result of obstructing ventilation on machinery that heats up, or flammable materials being too close to heaters.
Fuel can be anything that is combustible, such as wood, petroleum and spirits, and a number of gases. Solid fuels must reach a critical temperature in order to ignite, while many liquids release flammable vapours even when cold. Gases are the most hazardous and temperamental state, and can combust instantaneously.
The air feeding a fire only needs to be made up of 16% Oxygen in order to react with the heat and fuel. Generally, at low/normal altitude, the level is over 20%.
Effective Fire Extinction
If the three parts of the ‘fire triangle’ are kept in mind, extinguishing a small blaze should be a matter of common sense. The principles of fire extinction state that a fire will be put out if one of the three elements are removed, and this can be done using three different approaches, as detailed below.
Cooling
Removing the heat is one of the most effective methods of fire extinction available, which is why water is a popular extinguishing material. The fire will go out so long as the heat generated by the fire is less than that which is absorbed by the water.
Remember: water is not an appropriate extinguishing material to use on electrical fires, as well as those caused by cooking oils/fats or other flammable liquids.
Starving
While cooling removes the heat/ignition element of the ‘fire triangle’, starving the blaze of its fuel source approaches extinction from a different angle. A raging fire will burn itself out if it runs out of flammable materials, such as a bonfire out in the open that isn’t in contact with any other wood or dry grass. Similarly, a gas fire will immediately extinguish if the gas supply is cut off – you only have to look at a gas stove or Bunsen burner to see that.
Smothering
As the other key component present in the chemical reaction that causes combustion, removing oxygen from the equation is the final way of extinguishing a fire. For example, smothering a frying pan blaze with a fire blanket reduces the oxygen to below the 16% required to react, while covering a candle with a glass will snuff it out in a vacuum.
Smothering is a technique that is mostly applicable to solid fuel fires, although some materials may contain enough oxygen within their own chemical makeup to keep the blaze burning.