Fire is a process involving rapid oxidation at elevated temperatures accompanied by the evolution of heated gaseous products of combustion, and the emission of visible and invisible radiation.
Fire is a chemical process, which involves burning of any substance (combustion).
The combustible material that burns with the help of oxygen result in the production of heat & light, is called FIRE.
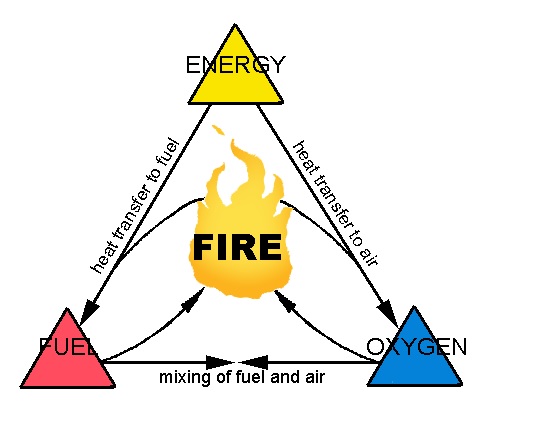
Oxygen + Heat + Fuel = Fire
In other words we can explain as :
Fire is a very rapid chemical reaction between oxygen and a combustible material, which results in the release of heat, light, flames, and smoke.
If You Discover a Fire Follow The 3 A’s
- Activate
- Assist
- Attempt
Stages of fire:-
Ignition: Fuel, oxygen and heat join together in a sustained chemical reaction. At this stage, a fire extinguisher can control the fire.
Growth: With the initial flame as a heat source, additional fuel ignites. Convection and radiation ignite more surfaces. The size of the fire increases and the plume reaches the ceiling. Hot gases collecting at the ceiling transfer heat, allowing all fuels in a room to come closer to their ignition temperature at the same time.
Fully developed: Fire has spread over much if not all the available fuel; temperatures reach their peak, resulting in heat damage. Oxygen is consumed rapidly.
Decay (Burnout): The fire consumes available fuel, temperatures decrease, fire gets less intense.
Explosion :-
Generally, An explosion is defined as a very rapid release of high-pressure gas into the environment. The energy from this very rapid release of the high-pressure gas is dissipated in the form of a shock wave.
Explosions can be classified as physical, a balloon bursting, as physical and/or chemical, a boiler explosion, or a chemical reaction of a gas/particle mixture. Our discussion will focus on chemical reaction explosions.
The process of a chemical reaction explosion is similar to the combustion process whereby a fuel and oxidant have premixed prior to ignition such as petroleum vapor or fine particles of grain dust mixed with air.
However, in an explosion the oxidation process proceeds at a greatly accelerated rate. The oxidation process is usually, but not always, confined within an enclosure such as a tank, grain silo, so that a rapid high-pressure rise occurs with an associated flame front. Generally, it is this high-pressure shock wave that causes the damaging effects from an explosion.


Leave a Comment